Introduction: While many publications have studied immune reconstitution after allogeneic stem cell transplantation (allo-SCT), data exploring whether the pre-transplant immune status (IS) of the recipients has any impact after this procedure are still lacking.
Material and Methods: Since May 2017, in our department, the IS of allo-SCT recipients has been systematically investigated at the time of pre-graft check-up. Data of this monocentric retrospective study on post-allo-SCT outcomes are reported here. Information was retrieved from complete blood counts (CBC) in terms of lymphocytes (normal range [NR] 1.5-4x109/L) and monocytes (NR: 0.15-0.9 x109/L). Classical multiparameter flow cytometry and a double platform were used to assess absolute counts of lymphocyte subsets, i.e. CD3+ (NR: 0.9-1.8 x109/L), CD4+ (NR: 0.5-1.2 x109/L), CD8+ (NR: 0.3-0.7 x109/L), CD19+ (NR: 0.1-0.4 x109/L) and CD56+ (NR: 0.1-0.4 x109/L). All patients also benefited from standard serum electrophoresis allowing to appreciate their global level of immunoglobulins (Ig) (NR: 8-13.6 g/L). The impact of pre-graft immune parameters was investigated for overall (OS) and disease-free (DFS) survivals as well as acute and chronic graft-versus-host disease (GVHD) incidence.
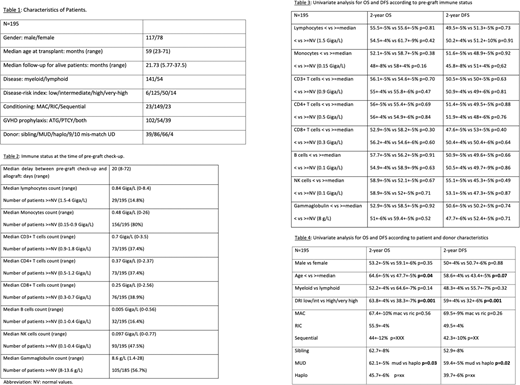
Results: Between May 2017 and December 2019, 195 consecutive adults were checked for immune status before allo-SCT. They were 117 males and 78 females at a median age of 59 years-old (range: 23-71). The median follow-up for the whole cohort was 21.7 months (range: 5.77-37.5). The majority of patients had a myeloid disease (n=141) and received a reduced intensity (RIC) regimen (n=149, myeloablative [MAC] n=23; sequential n=23). The disease-risk index was low/intermediate and high/very-high for respectively 91 and 104 patients. GVHD prophylaxis included anti-thymocytes globulins (ATG) for 102, post-transplant cyclophosphamide (PTCy) for 54 and both ATG+PTCy for 39. Donors were siblings in 39 cases, matched-unrelated (MUD) in 86, haplo-identical in 66 and 9/10 mis-matched in 4. The IS was evaluated at a median of 20 days (range: 8-72) before allo-SCT. Except for monocytes (median: 0.48 x109/L, range: 0-26), all median immune cell populations were lower than the NR before allo-SCT: lymphocytes (0.84 x109/L, range: 0-8.4), CD3+ (0.7 x109/L, range: 0-3.5), CD4+ (0.37 x109/L, range: 0-2.37), CD8+ (0.25 x109/L, range: 0-2.56), CD19+ (0.005 x109/L, range: 0-0.56), CD56+ (0.097 x109/L, range: 0-0.77). Conversely, the Ig median level was in the NR (8.6 g/L, range: 1.4-28). The percentages of patients with immune cells and Ig above the lowest NR were as follows: lymphocytes: 14.8%, monocytes: 80%, CD3: 37.4%, CD4: 37.4%, CD8: 38.9%, CD19: 16.4%, CD56: 47.6%, Ig: 56.7%. Only 5 patients (2.5%) had a fully normal IS before transplant. Considering the whole cohort, 2-year OS and DFS were 55.6+3% and 50.4+3%, respectively, while incidences of day 100 grade 2-4 and 3-4 acute GVHD and 2-year moderate/severe chronic GVHD were 34.7%, 19.4% and 19.8%, respectively.
The comparison of survivals between patients partitioned using either the median of the NR values for immune cells and Ig, showed no difference between subgroups. Similarly, IS had no impact on the incidence of acute nor chronic GVHD. Finally, no difference either was seen according to the conditioning regimen (RIC) or GVHD prophylaxis (ATG, PTCY or both).
Conclusion: As expected, almost all pre-ASCT patients present with immune depression. However, pre-transplant IS does not appear to impact survival nor GVHD. This suggests that there is no need to decrease or replace immunosuppressive treatments commonly used in recipients before allo-SCT in the hope to provide better outcomes.
Touzeau:GlaxoSmithKline: Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Other: Travel, Accommodations, Expenses; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Honoraria, Other: Travel, Accommodations, Expenses; Sanofi: Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Other: Travel, Accommodations, Expenses, Research Funding; Janssen: Consultancy, Honoraria, Other: Travel, Accommodations, Expenses. Le Gouill:Roche Genentech, Janssen-Cilag and Abbvie, Celgene, Jazz pharmaceutical, Gilead-kite, Loxo, Daiichi-Sankyo and Servier: Honoraria; Loxo Oncology at Lilly: Consultancy. Moreau:Abbvie: Consultancy, Honoraria; Novartis: Honoraria; Sanofi: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria; Takeda: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal